Approach to
Type 2 Diabetes Mellitus Management
Susan Joanne Wang1
Published online: 2 April 2021
1McGill University, Montreal, QC, Canada
Corresponding Author: Susan Joanne Wang, email susan.wang2@mail.mcgill.ca
DOI: 10.26443/mjm.v19i1.323
Abstract
Type 2 Diabetes Mellitus (T2DM) is a chronic and insidious disease that is on the rise worldwide. Diabetes pharmacotherapy is complex and varied, and recent studies of novel antihyperglycemic drugs have raised important considerations for the management of T2DM. This review provides an overview for the diagnosis of T2DM, glycemic targets for individuals with a T2DM diagnosis, and outlines a general approach to the management of T2DM, with an emphasis on how to select the appropriate pharmacotherapy using a fictional case as an example. Hypoglycemia, a complication of pharmacotherapy, macrovascular and microvascular disease resulting from T2DM, and other forms of diabetes mellitus are also briefly reviewed.
Tags: approach to, type 2 diabetes, guidelines, hyperglycemia, pharmacotherapy
Question
Mrs. S is a 34-year-old woman diagnosed with type 2 diabetes mellitus (T2DM) six months ago. At the time of diagnosis, her HbA1C was 7.4%. She started metformin and began walking for 30 minutes thrice a week. Three months later, her blood pressure (BP) was 125/80 mmHg, weight was 73.2 kg, and height was 161 cm (BMI 28.2); you increased the dose of metformin since her HbA1C was unchanged since her diagnosis. Today, her BP was stable, and she lost 2 kg, about which she is ecstatic. Her laboratory results are:
HbA1C: 7.3% (target in diabetes < 7.0%)
Plasma glucose, fasting: 9.2 mmol/L (normal: 3.0 - 6.1 mmol/L)
Creatinine: 58 µmol/L (normal: 53 - 106 µmol/L)
Estimated glomerular filtration rate (eGFR): 97 mL/min/1.73m2 (normal: ≥ 90 mL/min/1.73m2)
Triglycerides, fasting: 1.80 mmol/L (normal: < 1.70 mmol/L)
Total cholesterol, fasting: 6.56 mmol/L (normal: 3.00 - 5.20 mmol/L)
High-density lipoprotein (HDL), fasting: 1.22 mmol/L (normal: > 1.20 mmol/L)
Low-density lipoprotein (LDL), fasting: 2.11 mmol/L (target for diabetes < 2.00 mmol/L)
What is the next best step in managing this patient?
- Refer to a registered dietician (RD), start insulin therapy and follow-up in 3 months.
- Counsel on diet, start a sulfonylurea and follow-up in 3 months.
- Counsel on diet and increasing exercise, start insulin therapy and follow-up in 3 months.
- Refer to a RD, start a glucagon-like peptide-1 receptor agonist (GLP1-RA) and follow-up in 3 months.
- Refer to a RD, counsel on increasing exercise, start a sodium-glucose cotransporter-2 inhibitor (SGLT2i) and follow-up in 3 months.
Answer
E. Mrs. S is above her glycemic targets (HbA1C < 7.0% for adult diabetes). All patients with T2DM must be counselled on healthy behavior changes, and benefit from working with a RD and a personal trainer. Mrs. S lacks symptoms of hyperglycemia and does not need insulin therapy. Since she is happy with her weight loss, has an overweight BMI and elevated serum triglycerides and LDL, her weight and cardiovascular (CV) function are considered when selecting a drug.
Sulfonylureas are associated with weight gain and no CV benefits. GLP1-RAs and SGLT2is are associated with CV benefits and weight loss, but GLP1-RAs with CV benefits are injected while SGLT2is are given orally. Mrs. S should thus be counselled on exercise, referred to a RD and start a SGLT2i. A follow-up in 3 months is indicated to monitor therapeutic effectiveness, CV status and side effects.
Initial Approach
Diabetes mellitus is a chronic and insidious disease requiring frequent screening. Diabetes Canada has thorough guidelines with an approach to screening not covered here. (1) Evaluation of a patient with suspected T2DM begins with a complete history and physical examination. Patients can be asymptomatic, but signs and symptoms of hyperglycemia include polyuria, polydipsia, dehydration, blurry vision, headaches, fatigue, weakness, weight loss despite caloric intake, numbness or tingling in the extremities, and frequent infections. Cardiovascular disease (CVD) and microvascular disease affecting the eyes, kidneys and peripheral nerves are common at diagnosis.
Diagnosis
The diagnosis of diabetes is established by two confirmatory lab tests reflecting hyperglycemia or one lab test with symptoms of hyperglycemia. The diagnostic criteria are: 1) fasting plasma glucose (FPG) ≥ 7.0 mmol/L, where patients have no caloric intake for 8 hours preceding the test, 2) plasma glycated hemoglobin (HbA1C or A1C) ≥ 6.5%, considering factors affecting hemoglobin, 3) 2-hour plasma glucose (2hPG) in a 75 g oral glucose tolerance test (OGTT) ≥ 11.1 mmol/L), or 4) a random plasma glucose (PG) ≥ 11.1mmol/L, regardless of caloric intake timing (Flowchart 1). (2) Hemoglobinopathies and altered red cell turnover (e.g. hemolytic anemia) may affect A1C measurement.
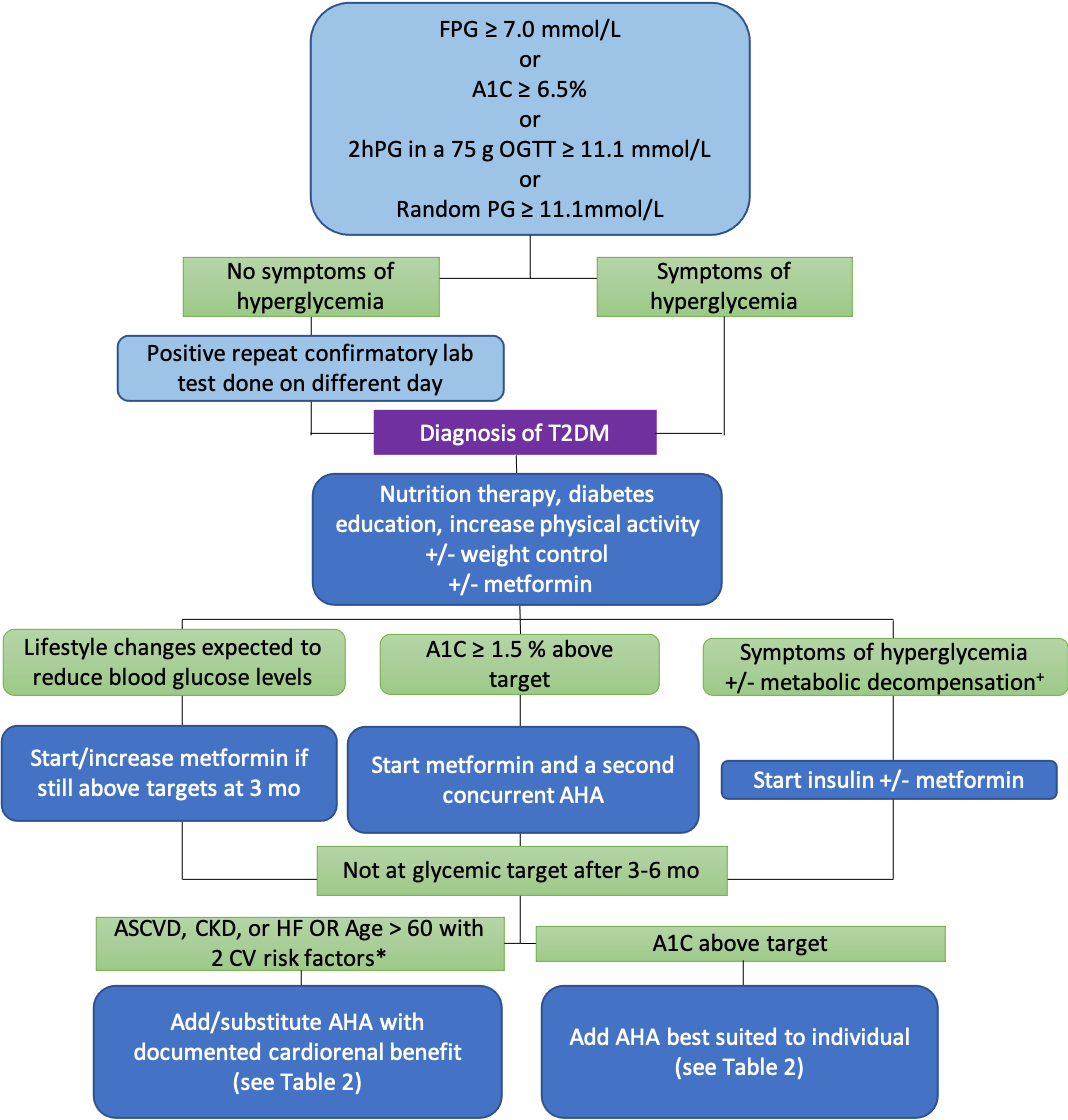
FPG = fasting plasma glucose; A1C = glycated hemoglobin; 2hPG = 2 hour plasma glucose; OGTT = oral glucose tolerance test; T2DM = type 2 diabetes mellitus; mo = months; AHA = antihyperglycemic agent; CKD = chronic kidney disease; HF = heart failure; CV = cardiovascular; ASCVD = atherosclerotic cardiovascular disease
+dehydration, diabetic ketoacidosis, hyperosmolar hyperglycemic state
*tobacco use; dyslipidemia (use of lipid-lowering agents, OR untreated low-density lipoprotein (LDL) ? 3.4 mmol/L, OR high-density lipoprotein (HDL) <1.0 mmol/L for men and <1.3 mmol/L for women, OR triglycerides >2.3 mmol/L); hypertension (use of antihypertensive therapy or untreated blood pressure ? 140/95); central obesity.
Adapted from: Pharmacologic Glycemic Management of Type 2 Diabetes in Adults 2020 Update, Canadian Journal of Diabetes. https://www.canadianjournalofdiabetes.com/action/showPdf?pii=S1499-2671%2820%2930228-8
Glycemic targets
Treatment of T2DM includes patient education, evaluation for macro- and microvascular complications, glycemic control, and reduction of cardiovascular risk factors and microvascular complications. (3,4) Targets for glycemic control are individualized depending on factors like age, kidney function, risk of hypoglycemia, functional dependence, and other comorbid conditions (Table 1). (1) In most cases, the target is an A1C ≤ 7.0%, which is correlated with reduced long-term micro- and macrovascular complications. (5,6)
| Table 1: Glycemic Control Targets for Type 1 and Type 2 Diabetes Mellitus | A1C% | Targets for Glycemic Control |
| ≤ 6.5 | Adults with diabetes to reduce risk of CKD and retinopathy if at low risk of hypoglycemia (for e.g. those with T2DM on oral hypoglycemic agents without risk of hypoglycemia). |
| ≤ 7.0 | Most adults with diabetes |
| 7.1 → 8.5 | Functionally dependent: 7.1-8.0% Recurrent severe hypoglycemia and/or hypoglycemia unawareness; limited life expectancy; frail elderly and/or with dementia: 7.1-8.5% |
| >8.5 | Avoid higher A1C to minimize risk of symptomatic hyperglycemia and complications |
Adapted from: Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada, Canadian Journal of Diabetes. http://guidelines.diabetes.ca/docs/CPG-2018-full-EN.pdf
Healthy behaviour interventions
Healthy behaviour interventions (or intensive lifestyle interventions (ILI)), such as diet, physical activity and smoking cessation, are a first-line intervention recommended in all patients with T2DM where reducing complications is a priority. (7) ILI implementation benefits from a multi-disciplinary approach, including RDs and certified fitness instructors. ILI is most effective in prediabetes or new-onset T2DM to reduce mortality and slow or reverse disease progression. (7) It can also contribute to a recommended 5-10% weight loss for overweight individuals, which can decrease insulin resistance, hyperglycemia, hypertension and dyslipidemia. (7,8)
Principles of diet in T2DM include choosing low glycemic index carbohydrates, reducing refined carbohydrates, increasing dietary fibre, and following the Eating Well with Canada's Food Guide. (9) Many dietary approaches, like the Mediterranean diet, have established benefits, but a patient's ethnocultural background should be considered. (10) Each diet must be individualized, regularly re-evaluated, and reinforced. It is also recommended that patients with T2DM reduce sedentary behaviour by engaging in 150 minutes of aerobic exercise and two resistance training sessions per week. (6,7)
First-line antihyperglycemic - Metformin
Due to the heterogeneity of T2DM, there are many classes of antihyperglycemics with varying mechanisms of lowering blood glucose (BG). Antihyperglycemics can be evaluated by characteristics according to the Safety, Tolerability, Effectiveness, Price, and Simplicity (STEPS) approach published by the American Academy of Family Physicians (Table 2). (11) Combining drugs with different mechanisms is common and preferred.
| Table 2: A STEPS Approach to Antihyperglycemic Agents | ||||||||
| Class | Mechanism | Safety | Tolerability | Effectiveness | Price | Simplicity | ||
| Cardio-renal | Weight (kg) | A1C | ||||||
| Biguanides metformin |
↑ insulin sensitivity by activating AMP kinase | Vitamin B12 deficiency, contraindicated in CKD, hold if risk of AKI | GI effects | ↓ CVD | neutral | 1.0% | $-$$ | Oral 1-2x/day |
| GLP-1 receptor agonists
-tide |
↑ glucose-dependent insulin release, slow gastric emptying, inhibit glucagon release | Contraindicated with Hx or FHx of medullary thyroid cancer or MEN 2, caution Hx of pancreatitis or pancreatic cancer, risk of retinopathy | GI effects, headache | ↓ CVD* | ↑ 1.1-4.4 | 0.6-1.4% | $$$$ | S.c. injection 2x/day, 1x/day or 1x/week |
| DPP-4 inhibitors -liptin |
Risk of HF with saxagliptin, caution Hx pancreatitis or pancreatic cancer | Rare: joint pain, pancreatitis | Neutral CVD | neutral | 0.5-0.7% | $$$ | Oral, 1x/day | |
| SGLT-2 inhibitors -liflozin |
Inhibits SGLT-2 to prevent glucose reuptake by kidney | Hypotension, rare DKA, caution with low carb eating or insulin deficiency, dapagliflozin contraindicated in bladder cancer, caution foot care (amputation risk), hold if risk of AKI | UTI, genital tract infections | ↓CVD
** ↓ renal disease*** |
↓ 2-3 | 0.5-0.7% | $$$ | Oral, 1x/day |
| Insulin | Activates insulin receptors | Hypoglycemia | Lipodystrophy | Neutral CVD | ↓ 1-3.5 | 0.9-1.2% or > | $ to $$$$ | S.c. injection 1-4x/day |
| Thiazolidinediones -glitazone |
↑ insulin sensitivity by activating peroxisome proliferator-activated receptors | CHF, fracture, possible increased risk MI with rosiglitazone, pioglitazone contraindicated in bladder cancer | Edema, rare: macular edema | neutral or ↑CVD | ↑2.0-2.5 | 0.7-0.9% | $$$ | Oral, 1x/day 6-12 weeks for max effect |
| α-glucosidase inhibitors acarbose |
Inhibits pancreatic α-amylase and intestinal α-glucosidase | Contraindicated in cirrhosis or CKD | GI effects | ? | neutral | 0.7-0.8% | $$ | Oral, 3x/day |
| Meglitinide repaglinide |
Activates sulfonylurea receptors on β-cell to stimulate insulin secretion | Hypoglycemia, contraindicated when combined with clopidogrel or gemfibrozil | GI effects, dizziness | ? | ↑ 1.4-3.3 | 0.7-1.1% | $$ | Oral, 3x/day |
| Sulfonylurea -zide, -ride |
Hypoglycemia, caution in G6PD deficiency | - | ? | ↑ 1.2-3.2 | 0.6-1.2% | $ | Oral, 1-2x/day | |
* no proven CV benefit with lixisenatide or short-acting exenatide; in established atherosclerotic cardiovascular disease (ASCVD), CKD OR >60 years with CV risk factors
** in established ASCVD, CKD, HF, OR >60 years with CV risk factors
*** in established ASCVD, CKD, OR >60 years with CV risk factors
Adapted from: Pharmacologic Glycemic Management of Type 2 Diabetes in Adults 2020 Update, Canadian Journal of Diabetes. https://www.canadianjournalofdiabetes.com/action/showPdf?pii=S1499-2671%2820%2930228-8 and Type 2 Diabetes Therapies: A STEPS Approach, American Academy of Family Physicians. https://www.aafp.org/afp/2019/0215/p237.html .
Biguanides, or metformin, are a class of antihyperglycemics that lower BG by activating AMP-activated protein kinase to enhance insulin sensitivity in the liver and peripheral tissues. (12) Metformin is a first-line antihyperglycemic and can be prescribed at diagnosis regardless of A1C level. It has a good safety profile with low risk of hypoglycemia when given as monotherapy, a neutral effect on weight, and is less expensive relative to other antihyperglycemics. (13) However, metformin is contraindicated in chronic kidney disease (CKD) with eGFR < 30 mL/min. (13)
Second-line antihyperglycemics
A patient with symptoms of hyperglycemia and/or metabolic decompensation, such as dehydration, weight loss, diabetic ketoacidosis (DKA), and a hyperosmolar hyperglycemic state, should be started on insulin immediately, regardless of A1C status. (13) A patient likely to respond to lifestyle changes will require metformin initiation or escalation if they have not attained targets within 3 months (Flowchart 1). A non-insulin and non-metformin antihyperglycemic should be considered in all patients who do not attain targets within 3 to 6 additional months. A patient with an A1C ≥ 1.5% above target should be concurrently started on metformin and a second antihyperglycemic. (13)
In the presence of atherosclerotic cardiovascular disease (ASCVD), CKD, heart failure or an age > 60 years, and two CV risk factors (Flowchart 1), patients should start antihyperglycemics with demonstrated cardiorenal benefits. (14) These include SGLT2is, such as empagliflozin and canagliflozin, and GLP1-RAs, such as liraglutide, semaglutide or dulaglutide. Patients without ASCVD, CKD or risk factors can start an additional antihyperglycemic according to their clinical needs (Table 2). (11,14) If avoidance of weight gain and hypoglycemia are priorities, dipeptidyl peptidase-4 inhibitors (DPP-4is), GLP1-RAs and/or SGLT2is are recommended.
The A1C value tends to decrease by 0.5% to 1.5% on monotherapy, usually achieving maximal effects by 3 to 6 months. (15) Beta cell function declines over time, often leading BG levels to rise insidiously despite treatment adherence; thus, treatment regimens are frequently adjusted and tailored.
Insulin therapy
Insulin therapy is effective for individuals with significant hyperglycemia and can lead to partial recovery of beta cell function in patients with metabolic decompensation. (13,16) Insulin is primarily delivered via injection and is rarely associated with lipodystrophy at the injection site. Long-acting or intermediate-acting insulin analogue injections are used for basal glycemic control while bolus injections at mealtimes are used for prandial glycemic control. Combining insulin with other antihyperglycemics can lead to better glycemic control with less insulin and fewer side effects compared to insulin alone. (13) Risk of hypoglycemia is high with insulin; thus, regimens must be adjusted to reduce risk, especially in the elderly. Reduction of A1C is directly correlated with the dose and number of daily injections.
Beyond the Initial Approach
This section covers special considerations for further evaluation and management of T2DM, as well as distinctions from other forms of diabetes mellitus.
Hypoglycemia
Hypoglycemia is defined by a plasma glucose of < 4 mmol/L and the presence of autonomic or neuroglycopenic symptoms that resolve with the administration of carbohydrates. Some symptoms include trembling, palpitations, sweating, anxiety, nausea, confusion, weakness, and vision changes. Hypoglycemia is potentially dangerous if it occurs while driving or operating machinery. Prolonged hypoglycemia can result in coma, paresis, convulsion, and encephalopathy, while repeated episodes can lead to hypoglycemia unawareness and mild intellectual impairment. (17) The best management for hypoglycemia is prevention and patients must be counselled on how to recognize and manage symptoms. This includes awareness of BG levels, especially before driving, as well as preparing and carrying 20 g of fast-acting sugar. (17
Cardiovascular disease
Diabetes significantly accelerates the natural history and development of CVD; therefore, special attention must be given to CV health in T2DM. BP should be < 130/80 mmHg and LDL < 2.0 mmol/L or a > 50% reduction from baseline. (18) Additional pharmacotherapy may be initiated in individuals with high risk or presence of CVD. Drugs include angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers (ARBs), statins, aspirin, GLP1-RAs and SGLT2is. Statins are indicated in patients with clinical CVD and 1) age ≥ 40 years, or 2) age > 30 with diabetes > 15 years, or 3) age < 40 with microvascular disease. (18)
Microvascular disease
Annual screening is critical to detect and treat microvascular complications like diabetic retinopathy, neuropathy, and nephropathy. In all cases, BG and BP management is the best way to prevent or slow progression of these pathologies. Referral to a specialist is usually warranted. Retinopathy may be treated by laser therapy or vitrectomy and may benefit from statins or fenofibrate. (19) Neuropathy is screened by 10 g monofilament or vibration perception tests and managed with analgesics or other medications like anti-depressants. CKD is defined as an eGFR < 60 mL/min or the presence of microalbuminuria or proteinuria (a random urine albumin to creatinine ratio ≥ 2.0 mg/mmol in ≥ 2 of 3 samples over 3 months). Low eGFR is associated with a high risk of CVD. ACE inhibitors, ARBs and SLGT2is should be considered for the management of BP and to slow the progression of CKD. (19) SGLT2is have been shown to be effective in early CKD. (14)
Other forms of diabetes mellitus
Although T2DM is the most common form of diabetes mellitus, its management differs greatly from that of gestational diabetes mellitus (GDM) and type 1 diabetes mellitus (T1DM). GDM, defined as diabetes with onset during pregnancy, is screened for during the second trimester. First-line therapy focuses on healthy behaviour interventions, followed by insulin +/- metformin if targets are not met. (20) T1DM is an autoimmune condition where pancreatic beta cells are attacked by one's own immune system, resulting in the absence of insulin production. Exogenous insulin therapy is therefore required for these patients to avoid metabolic decompensation. Diagnosis of T1DM usually occurs in the first two decades of life; management of hyperglycemia requires continuous BG monitoring and insulin delivery, either by insulin pump or multiple daily injections.
References
- Ekoe J, Goldenberg R, Katz P. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Screening for Diabetes in Adults. Can J Diabetes 2018;42(Suppl 1):S16-S19. https://guidelines.diabetes.ca/cpg/chapter4
- Punthakee Z, Goldenberg R, Katz P. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Definition, Classification and Diagnosis of Diabetes, Prediabetes and Metabolic Syndrome. Can J Diabetes 2018;42(Suppl 1):S10-S15. https://guidelines.diabetes.ca/cpg/chapter3
- Pinsker JE, Shank T, Dassau E, Kerr D. Comment on American Diabetes Association. Approaches to Glycemic Treatment. Sec. 7. In Standards of Medical Care in Diabetes-2015. Diabetes Care 2015; 38 (Suppl. 1): S41-S48. Diabetes Care. 2015 Oct 1;38(10):e174-e174. doi: 10.2337/dc15-0839
- Imran SA, Agarwal G, Bajaj HS, Ross S. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Targets for Glycemic Control. Can J Diabetes 2018;42(Suppl 1):S42-S46. https://guidelines.diabetes.ca/cpg/chapter8
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). The Lancet. 1998 Sep 12;352(9131):837-53. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736%2898%2907019-6/fulltext
- Writing Team for the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Effect of intensive therapy on the microvascular complications of type 1 diabetes mellitus. JAMA 2002;287:2563-9. 10.1001/jama.287.19.2563
- Diabetes Prevention Program Research Group. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. The Lancet. 2009 Nov 14;374(9702):1677-86. doi: 10.1016/S0140-6736(09)61457-4
- Look AHEAD Research Group, Wing RR, Bolin P, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes [published correction appears in N Engl J Med. 2014 May 8;370(19):1866]. N Engl J Med. 2013;369(2):145-154.
- Ricci-Cabello I, Ruiz-Pérez I, Rojas-García A, Pastor G, Rodríguez-Barranco M, Gonçalves DC. Characteristics and effectiveness of diabetes self-management educational programs targeted to racial/ethnic minority groups: a systematic review, meta-analysis and meta-regression. BMC endocrine disorders. 2014 Dec 1;14(1):60.
- Marcy TR, Britton ML, Harrison D. Identification of barriers to appropriate dietary behavior in low-income patients with type 2 diabetes mellitus. Diabetes Ther 2011;2:9-19.
- Steinberg J, Carlson L. Type 2 Diabetes Therapies: A STEPS Approach. American family physician. 2019 Feb 15;99(4):237-43. https://www.aafp.org/afp/2019/0215/p237.html
- Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N. Role of AMP-activated protein kinase in mechanism of metformin action. The Journal of clinical investigation. 2001 Oct 15;108(8):1167-74. https://www.jci.org/articles/view/13505?content_type=full
- Lipsombe L, Booth G, Butalia S, Dasgupta K, et al. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Pharmacologic Glycemic Management of Type 2 Diabetes in Adults. Can J Diabetes 2018;42(Suppl 1):S88-S103.https://guidelines.diabetes.ca/cpg/chapter13
- Lipscombe L, Butalia S, Dasgupta K, Eurich DT, MacCallum L, Shah BR, Simpson S, Senior PA. Pharmacologic glycemic management of type 2 diabetes in adults: 2020 update. Canadian Journal of Diabetes. 2020 Oct 1;44(7):575-91.https://doi.org/10.1016/j.jcjd.2020.08.001
- Sherifali D, Nerenberg K, Pullenayegum E, Cheng JE, Gerstein HC. The effect of oral antidiabetic agents on A1C levels: a systematic review and meta-analysis. Diabetes care. 2010 Aug 1;33(8):1859-64.
- Weng J, Li Y, Xu W, Shi L, Zhang Q, Zhu D, Hu Y, Zhou Z, Yan X, Tian H, Ran X. Effect of intensive insulin therapy on ?-cell function and glycaemic control in patients with newly diagnosed type 2 diabetes: a multicentre randomised parallel-group trial. The Lancet. 2008 May 24;371(9626):1753-60. https://europepmc.org/article/med/18502299
- Yale J, Paty P, Senior, PA. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Hypoglycemia. Can J Diabetes 2018;42(Suppl 1):S104-S108.https://guidelines.diabetes.ca/cpg/chapter14
- Stone JA, Houlden RL, Lin P, Udell JA, Verma S. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Cardiovascular protection in People with Diabetes. Can J Diabetes 2018;42(Suppl 1):S162-S169.https://guidelines.diabetes.ca/cpg/chapter23
- Stone JA, Houlden RL, Lin P, Udell JA, Verma S. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Cardiovascular protection in People with Diabetes. Can J Diabetes 2018;42(Suppl 1):S201-S209.https://guidelines.diabetes.ca/cpg/chapter29
- Feig DS, Murphy K, Asztalos E, Tomlinson G, Sanchez J, Zinman B, Ohlsson A, Ryan EA, Fantus IG, Armson AB, Lipscombe LL. Metformin in women with type 2 diabetes in pregnancy (MiTy): a multi-center randomized controlled trial. BMC pregnancy and childbirth. 2016 Dec;16(1):1-8.

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.